UK first in world to start using Oxford-AstraZeneca vaccine
The U.K. on Monday became the first nation in the world to start using the COVID-19 vaccine developed by Oxford University and drugmaker AstraZeneca, ramping up a nationwide inoculation program as rising infection rates are putting an unprecedented strain on British hospitals.
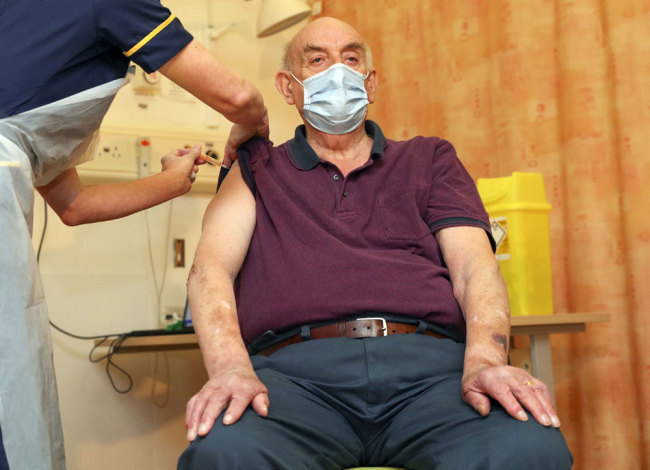
82-year-old Brian Pinker receives the Oxford University/AstraZeneca COVID-19 vaccine from nurse Sam Foster at the Churchill Hospital in Oxford, England, Monday, Jan. 4, 2021. [Photo: Pool via AP/ Steve Parsons]
Brian Pinker, an 82-year-old dialysis patient, received the first shot at 7:30 a.m. at Oxford University Hospital.
"The nurses, doctors and staff today have all been brilliant, and I can now really look forward to celebrating my 48th wedding anniversary with my wife, Shirley, later this year," Pinker said in a statement released by the National Health Service.
The rollout of the new vaccine comes at a crucial moment for U.K. authorities, who are battling a surge in infections blamed on a new virus variant that authorities have said is much more contagious. Prime Minister Boris Johnson, who announced a wave of tight restrictions the weekend before Christmas, says even tougher regulations will be announced soon.
The U.K. is in the midst of an acute outbreak, recording more than 50,000 new coronavirus infections a day over the past six days. On Sunday, it notched up another 54,990 cases and 454 more virus-related deaths to take its confirmed pandemic death toll total to over 75,000, one of the worst in Europe. Some areas northeast of London have infection rates of over 1,000 cases per 100,000 people.

Britain's Prime Minister Boris Johnson watches as nurse Jennifer Dumasi is injected with the Oxford-AstraZeneca Covid-19 vaccine, during a visit to view the vaccination programme at the Chase Farm Hospital in north London, Monday Jan. 4, 2021, part of the Royal Free London NHS Foundation Trust. [Photo: Pool via AP/Stefan Rousseau]
"If you look at the numbers, there's no question we will have to take tougher measures and we will be announcing those in due course," Johnson said Monday.
U.K. regulators last week authorized emergency use of the Oxford-AstraZeneca shot, giving public health officials a second vaccine in their medical arsenal. Britain's mass vaccination program began Dec. 8 with the shot developed by New York-based Pfizer and its German partner BioNTech.
Britain has secured the rights to 100 million doses of the Oxford-AstraZeneca vaccine, which is cheaper and easier to use than some of its rivals. In particular, it doesn't require the super-cold storage needed for the Pfizer vaccine.
The new vaccine will be administered at a small number of hospitals for the first few days so authorities can watch out for any adverse reactions. But the NHS said hundreds of new vaccination sites — including local doctors' offices — will open later this week, joining the more than 700 vaccination sites already in operation.
A "massive ramp up operation" is now underway in the vaccination program, Johnson said Monday at Chase Farm Hospital in north London, where he met with some of the first people to receive the Oxford-AstraZeneca shot.

Chief nurse Sam Foster prepares to administer a dose of the Oxford University/AstraZeneca COVID-19 vaccine, at the Churchill Hospital in Oxford, England, Monday, Jan. 4, 2021. [Photo: Pool via AP/ Steve Parsons]
But aspects of Britain's vaccination plans have spurred controversy.
Both vaccines require two shots, and Pfizer had recommended that the second dose be given within 21 days of the first. But The U.K.'s Joint Committee on Vaccination and Immunization said authorities should give the first vaccine dose to as many people as possible, rather than setting aside shots to ensure others receive two doses. It has stretched out the time between the doses from 21 days to within 12 weeks.